Qu'est-ce que SUDOSCAN ?
SUDOSCAN est un dispositif unique et innovant. Il s'agit d'une technologie simple et non invasive, capable de détecter et de suivre les neuropathies à petites fibres qui ne sont généralement pas détectées par les outils traditionnels.
Comment utiliser SUDOSCAN ?
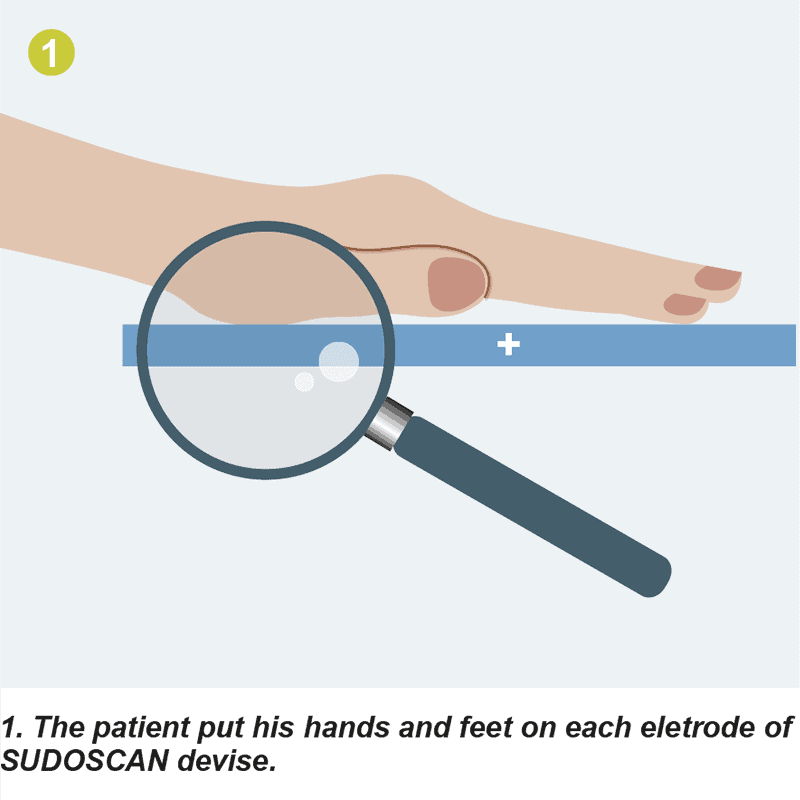
L'appareil se compose d'un ordinateur et de 4 électrodes sur lesquelles les patients placent leurs mains et leurs pieds nus. En moins de 3 minutes, SUDOSCAN propose une stimulation des glandes sudoripares qui évalue de petites fibres nerveuses (fibres C). Cette détection précoce améliore la prise en charge de la maladie et permet d'éviter les complications sévères associées à certaines maladies.
SUDOSCAN Principe
SUDOSCAN mesure la capacité des glandes sudoripares à libérer des ions chlorure en réponse à un stimulus électrique sur la paume des mains et la plante des pieds, zones où la densité des glandes sudoripares est la plus élevée.
L'utilité de SUDOSCAN a été démontrée dans :
- Neurologie: pour l'évaluation et le suivi de pathologies telles que la gestion de la douleur neuropathique, les neuropathies idiopathiques, le Gougerot Sjögren, l'amyloïdose, la maladie de Fabry, etc.
- Diabétologie: pour la détection précoce de la neuropathie périphérique et du dysfonctionnement des glandes sudoripares, utile pour la prévention des complications du pied diabétique.
- Oncologie: pour une évaluation quantitative de la neuropathie induite par la chimiothérapie, permettant une décision meilleure et informée.
- Suivi: pour la gestion des patients, le suivi longitudinal des traitements et les interventions sur le mode de vie.
Une technologie de confiance
SUDOSCAN a des résultats similaires à ceux des tests de référence tels que la biopsie cutanée, le QSART ou les scores cliniques comme le NIS-LL. La technologie est étayée par plus de 150 publications dans des revues scientifiques internationales.
Le dispositif est fabriqué en France et le test est très sensible et hautement reproductible.
SUDOSCAN est certifié CE, autorisé par la FDA américaine, et est enregistré et distribué dans 34 pays avec plus de 5000 appareils vendus.
SUDOSCAN mesure la capacité des glandes sudoripares à libérer des ions chlorure en réponse à un stimulus électrique sur la paume des mains et la plante des pieds, zones où la densité des glandes sudoripares est la plus élevée :
 Diabète [Prévenir et gérer les complications du diabète].
Diabète [Prévenir et gérer les complications du diabète].
Complications du diabète
- L'efficacité de SUDOSCAN dans la détection précoce de la neuropathie diabétique a été démontrée dans plusieurs études.
- SUDOSCAN permet également une meilleure prise en charge des complications du diabète telles que le pied diabétique et la neuropathie cardiaque autonome, ainsi que le suivi de l'évolution de la maladie.
> Pour en savoir plus, consultez notre section Sudoscan et diabète.
 Neurologie
Neurologie
Maladies avec complications neurologiques
SUDOSCAN a été appliqué à plusieurs maladies ayant des complications neurologiques, telles que l'amylose héréditaire, les neuropathies idiopathiques, le Covid-19, la maladie de Fabry, la maladie de Parkinson, la neuropathie auto-immune à petites fibres, la fibromyalgie, le syndrome de Sjörgen, la défaillance autonome pure, l'hépatite C.
>Pour en savoir plus, consultez notre section Sudoscan en neurologie.
 Oncologie
Oncologie
Neuropathie périphérique induite par la chimiothérapie
SUDOSCAN peut être facilement réalisé dans le service d'oncologie, avant et après le traitement, pour un suivi optimal des patients atteints de polyneuropathie induite par la chimiothérapie (CIPN). Les résultats de SUDOSCAN sont corrélés à l'évaluation clinique du Total Neuropathy Score (TNSc) : réduit pour TNSc > 6 population.
>Pour en savoir plus, consultez notre section Sudoscan en oncologie.
 Suivi
Suivi
Suivi des résultats pour les patients
Une autre application clé de SUDOSCAN est la gestion des patients, le suivi longitudinal des traitements et l'impact sur le mode de vie. SUDOSCAN a été utilisé pour évaluer l'effet de l'activité physique sur le risque cardiovasculaire et les maladies métaboliques.
>Pour en savoir plus, consultez notre section Suivi avec Sudoscan.
Développement futur
SUDOSCAN est impliqué dans le projet européen e-PREDICE (Early PRevention of Dlabetes Complications in people with hyperglycaemia in Europe). e-PREDICE a pour but de tester l'efficacité à long terme de différents schémas thérapeutiques pour la prévention précoce des complications diabétiques chez les personnes souffrant d'hyperglycémie et soumises à un mode de vie actif. Ce projet impliquant 15 centres en Europe a été développé en coopération avec le Pr. Jaakko TUOMILEHTO.
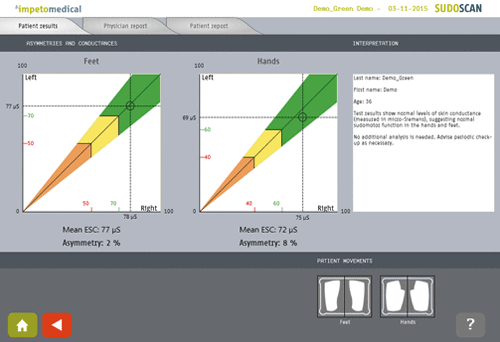
Les résultats du test SUDOSCAN peuvent servir d'indicateurs pour les patients présentant un risque de dysfonctionnement autonome grâce à la mesure de Conductance Cutanée Electrochimique (CCE) et à la quantité d'asymétrie observée entre le côté droit et le côté gauche.
Un test de la fonction sudorale
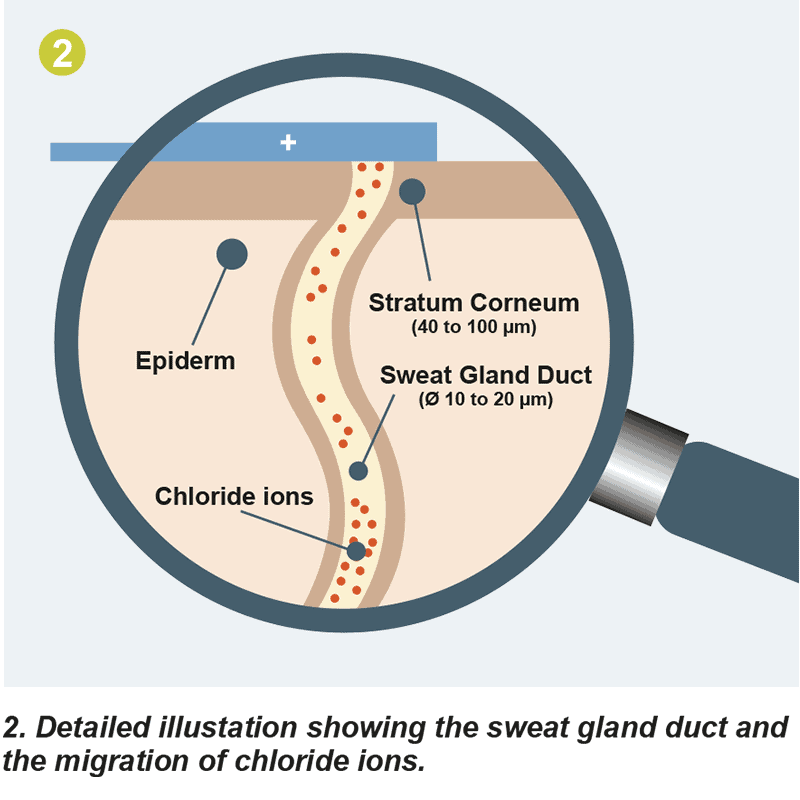
SUDOSCAN teste le fonctionnement des glandes sudoripares en appliquant un petit courant continu à la fois sur les électrodes de mains et de pieds. À basse tension, la couche cornée agit comme un condensateur, laissant les canaux sudoripares comme seuls canaux pour la transmission des ions Cl-.
SUDOSCAN fournit une mesure quantitative de la conductance du chlorure (mesurée en microsiemens) et ses résultats servent de biomarqueur pour évaluer la fonction des glandes sudoripares par rapport à leur innervation.
Que se passe-t-il pendant un examen ?
Pendant le scanner, l'appareil envoie un petit courant électrique à travers les électrodes. Ce courant est très faible, inférieur à 4V, et n'est pas douloureux pour le patient. En revanche, ce courant est considéré comme très fort pour les glandes sudoripares. Il les oblige à réagir en libérant des ions chlorure, naturellement présents dans les glandes sudoripares saines.
Après 3 minutes, les résultats quantitatifs et objectifs pour les deux mains et les deux pieds s'affichent immédiatement pour aider à évaluer les petites fibres C. Ce test est un moyen simple, rapide et non invasif de détecter la neuropathie autonome périphérique à petites fibres.
Confirmé par la science
Comme le test est approuvé par des leaders d'opinion mondiaux, la sensibilisation à cette technologie augmente rapidement. SUDOSCAN est en train de devenir un test de référence dans de nombreux pays. Il a notamment été inclus dans différentes directives telles que le réseau européen pour l'amylose TTR-FAP, les directives allemandes pour la gestion du diabète ou l'association latino-américaine du diabète. Il est commercialisé dans le monde entier avec plus de 3000 appareils vendus. SUDOSCAN est certifié CE, autorisé à l'utilisation par la FDA américaine, et est enregistré et distribué dans 34 pays.
Comment utiliser SUDOSCAN ?
SUDOSCAN est un test qui permet une évaluation précise de la fonction sudorale. Le test se concentre sur les petites fibres nerveuses du système nerveux périphérique qui innervent les glandes sudoripares.
L'appareil se compose d'un ordinateur et de 4 électrodes sur lesquelles les patients placent leurs mains et leurs pieds nus. En moins de 3 minutes, SUDOSCAN offre une stimulation des glandes sudoripares qui évalue les petites fibres nerveuses (fibres C).
Des résultats simples obtenus en 3 minutes
- Les résultats s'affichent automatiquement.
- Si le score est élevé, cela signifie qu'il n'y a pas de signe de dysfonctionnement sudomoteur ni de neuropathie autonome.
- Si le score est faible, cela indique un dysfonctionnement sudomoteur et une neuropathie.
Des résultats faciles à lire
La polarité des plaques de capteurs en acier inoxydable est inversée pendant le balayage afin d'enregistrer les valeurs de conductance des côtés droit et gauche. Les résultats du patient sont affichés dès la fin de l'examen. L'importance de l'asymétrie observée est un indicateur du type de neuropathie périphérique.
Le médecin peut également afficher la courbe de suivi dans le temps de plusieurs scans SUDOSCAN.
Résultats SUDOSCAN comparés à la biopsie cutanée et au QSART
L'efficacité de SUDOSCAN a été comparée aux principales techniques d'évaluation des neuropathies à petites fibres dans plusieurs publications scientifiques. G. Smith et al [1] ont démontré que les performances diagnostiques de SUDOSCAN étaient comparables à celles de l'Intraepidermal Nerve Fiber Density (IENFD) et du QSART dans la détection de la neuropathie.
Les volumes de sueur de la jambe distale et du pied QSART de l'IENFD avaient une aire sous la courbe (AUC) similaire aux valeurs de SUDOSCAN (ESC).
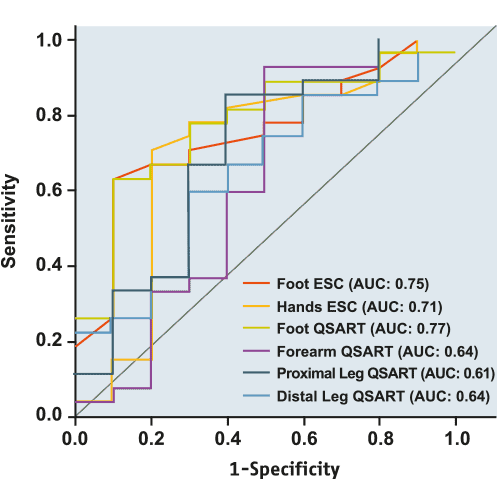
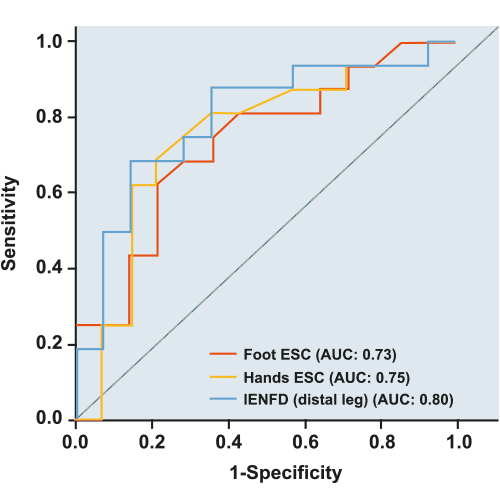
IENFD : densité des fibres nerveuses intraépidermiques obtenue à partir d'une biopsie cutanée.
QSART : Quantitative Sudomotor Axon Reflex Testing - test de la fonction sudomotrice.
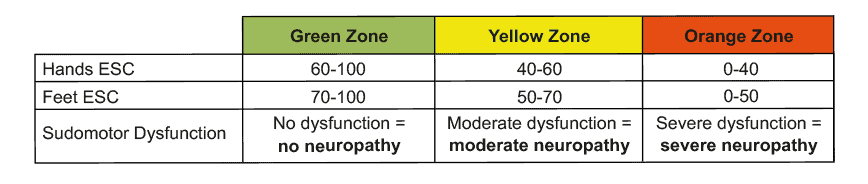
Valeurs de référence évaluées dans une large population d'adultes
Une large population d'adultes a été testée pour la neuropathie avec SUDOCAN [2]. Des valeurs de référence ont ainsi été déterminées à trois niveaux : conductance électrochimique de la peau (CCE) pour une fonction sudorale normale (vert), un dysfonctionnement sudoral détecté (jaune) et un dysfonctionnement sudoral grave (rouge).
Références :[1] Smith G et al. The diagnostic utility of Sudoscan for distal symmetric peripheral neuropathy. Journal of Diabetes and its Complications. 2014;28(4):511-516.[2] Vinik AI, Nevoret ML, Casellini C . Le nouvel âge du test de la fonction sudomotrice : A Sensitive and Specific Biomarker for Diagnosis, Estimation of Severity, Monitoring Progression, and Regression in Response to Intervention. Front Endocrinol (Lausanne). 2015 Jun 11;6:94.
 RAPIDE
RAPIDE
- Pas de préparation du patient
- Résultats en 3 minutes
- Points de données critiques faciles à lire pour aider les médecins à établir un diagnostic
 SÉCURITÉ
SÉCURITÉ
- Non invasif
- Pas de jeûne
- Facile à utiliser
- Homologations CE et FDA
 PRÉCIS
PRÉCIS
- Des résultats quantitatifs reproductibles
- Indépendant des conditions environnementales
- Soutenu par des recherches fondées sur des données probantes
- 270 publications dans des revues à comité de lecture